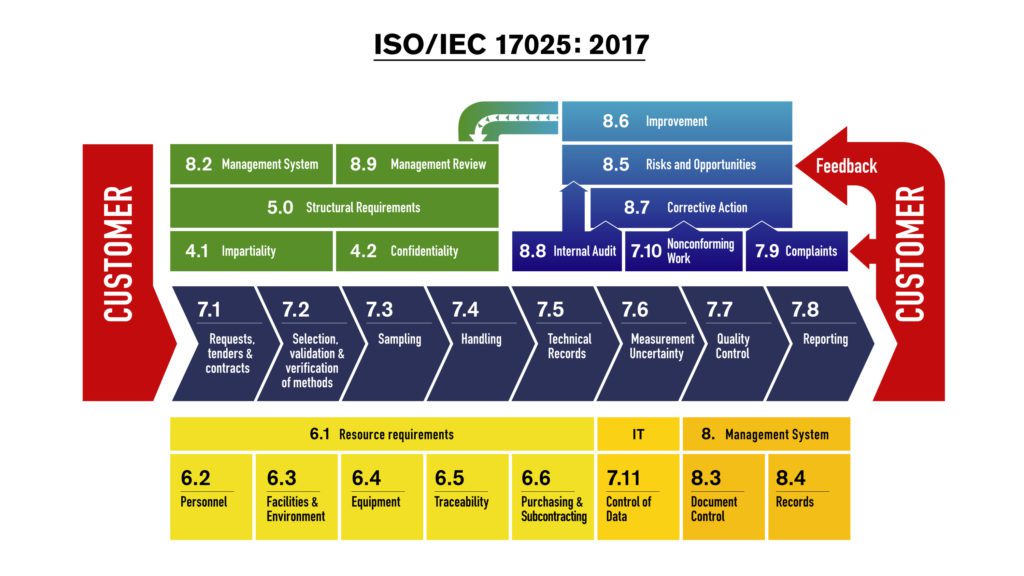
The Selection, Verification and Validation of Methods is one of the most technical and most important parts of the standard. The section begins with the Review of Requests, Tenders and Contracts. This section covers 11 core processes to improve efficiency. There are six clauses that address the requirement for the laboratory to have available the personnel, facilities, equipment, systems, and support services necessary to perform its laboratory activities. Section 5 also defines the basic requirements for personnel, the authority given to them, and the resources needed to carry out their duties. Section 5 sets management’s responsibilities in an accredited laboratory and their responsibilities to customers, regulatory authorities, and organizations that provide recognition. It states that an accredited laboratory must be a legal entity or part of a legal entity, which is responsible for its testing and calibration activities. This section defines the basic organizational components of a laboratory, its range of activities, and its commitment to an effective management system. Confidentially requires the laboratory to keep all results and information private. Internal issues, personal relationships, or other conflicts of interest are addressed and resolved. Impartiality implies that the laboratory will not allow commercial, financial, or other pressures to compromise the quality of results. This section covers impartiality and confidentiality, two requirements that are vital for maintaining the trust and confidence that the users of tests and calibrations place in the laboratories they use. The ISO/IEC 17025:2017 structure is divided into five main sections that contain the requirements for laboratory accreditation. ISO/IEC 17025 assists in the harmonization of procedures and standard methods, facilitates cooperation between laboratories and other bodies, and promotes the acceptance of results between countries. Laboratory sampling, testing, and conformity assessment are crucial enabling factors for technology and trade.
Iso 17025 2017 iso#
Getting to the heart of why ISO 17025 is important For example, calibration can be used to ensure that the scale at the airport (the unknown) will accurately weigh your luggage by comparing the readings across against certified mass pieces (the known). Calibration laboratories, on the other hand, compare a measurement instrument of unknown accuracy to one of known accuracy. An example is testing a cereal sample (the item of interest), to see if the amount of pesticides it contains meets the legislated limits (the conformity). Testing laboratories determine the characteristics of an item of interest for conformity assessment. What are testing and calibration laboratories? ISO/IEC 17025, together with ISO 9001, is the basis for ISO 15189, which specifies particular requirements for competence and quality, as well as accreditation of medical laboratories. This includes regulated government and public sectors, as well as non-regulated sectors. The standard and accreditation are used by independently owned and operated laboratories, as well as those that are part of larger organizations, irrespective of the industry and size, which are involved in sampling or measurement activities. Accreditation is a formal declaration by an Accreditation Body, after assessment and confirmation, that a laboratory is effective in meeting the requirements of ISO 17025 to perform tests according to its accredited scope. Whereas certification according to ISO 9001 or ISO 14001 is recognition that an effective management system is in place, ISO/IEC 17025 includes recognition of the technical competence of laboratories. The standard was published with collaboration between the International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC). ISO/IEC 17025:2017 is the current, revised standard.

It specifies the activities that must be included in laboratory operations to promote confidence in its ability to produce valid and consistently reliable testing, calibration, and sampling results. ISO/IEC 17025 is the international standard that sets out the general requirements for the competent, impartial, and consistent operation of laboratories.

A simple introduction to the basics of ISO/IEC 17025 This introductory guide provides insight into how ISO 17025, the main standard for laboratories, can help your company, why it is important, its basic layout, and what the implementation entails. “What is ISO/IEC 17025:2017?” Simple answers for a complex subject
